CRISPR-based genome-editing therapies hold immense potential, but their reliance on bacterial proteins called CRISPR Nucleases can trigger human immune responses. This immune activation poses risks of side effects and diminishes the effectiveness of the therapies. In collaboration with Cyrus Biotechnology, researchers from the Broad Institute of MIT and Harvard have developed engineered versions of two widely used CRISPR nucleases—Cas9 and Cas12—that evade immune detection while maintaining their gene-editing efficiency.
The breakthrough study, published in Nature Communications, was led by Feng Zhang, a prominent figure in genome editing. Zhang’s team pinpointed the protein sequences on Cas9 and Cas12 that provoke immune reactions, using computational tools to redesign the nucleases to avoid immune recognition.
Targeting the Root of Immune Reactions: CRISPR Nucleases

Cas9 and Cas12 nucleases, commonly derived from bacteria like Streptococcus pyogenes and Staphylococcus aureus, are central to many CRISPR therapies. However, about 80% of people have pre-existing immunity to these bacterial proteins due to natural exposure, increasing the likelihood of immune responses during treatment.
The research team used mass spectrometry to identify specific fragments—short sequences of about eight amino acids—that were recognized by the immune system. These sequences acted as the immune system’s targets, sparking unwanted reactions.
By partnering with Cyrus Biotechnology, Zhang’s team employed computational modeling to redesign the nucleases, removing the immune-triggering sequences while preserving their ability to cut DNA.
Testing the Engineered Nucleases: CRISPR Nucleases
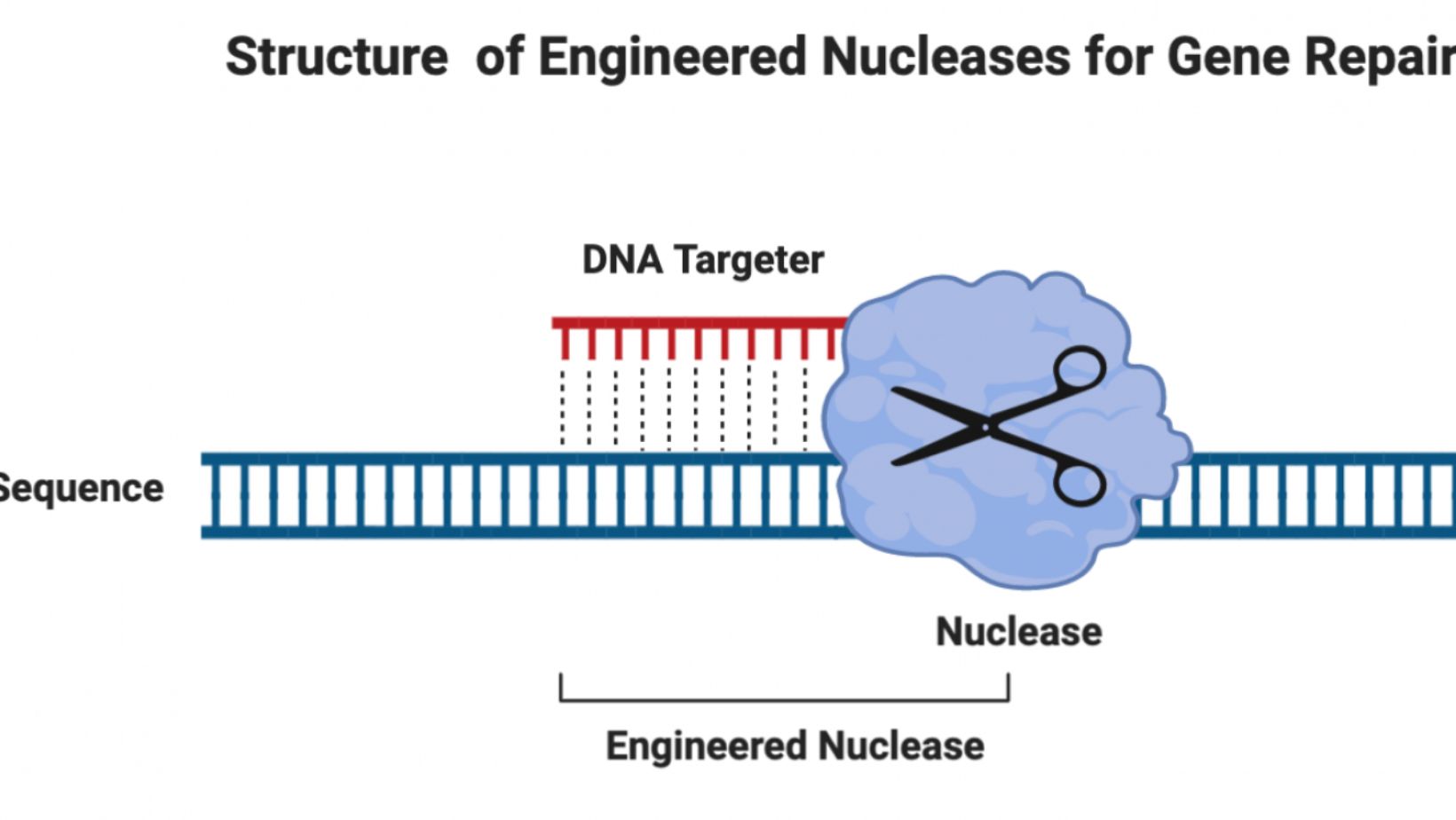
To confirm the effectiveness of the redesigned nucleases, the researchers tested them in both human cells and mice genetically modified with human immune system components. The results were promising: the engineered enzymes triggered significantly lower immune responses but retained their gene-editing efficiency.
“People have known for a while that Cas9 causes an immune response,” said Rumya Raghavan, co-first author of the study. “We wanted to pinpoint which parts of the protein were being recognized by the immune system and then engineer the proteins to get rid of those parts while retaining their function.”
Toward Safer and More Effective Gene Therapies
Developing minimally immunogenic nucleases is a significant step toward safer gene-editing therapies. “Our goal was to create not only a safer therapy but one potentially more effective because the immune system is not eliminating it before it can do its job,” said Mirco Julian Friedrich, co-first author.
While these engineered nucleases address one challenge of the immune response, Zhang’s team sees this as part of a larger goal to refine gene therapies. Future research aims to design delivery vehicles capable of evading the immune system, further enhancing the safety and efficiency of CRISPR treatments.
This innovation represents a pivotal advance in making genome-editing therapies more accessible and safer, opening the door to their broader application in treating genetic diseases.
Reference: Rumya Raghavan et al, Rational engineering of minimally immunogenic nucleases for gene therapy, Nature Communications (2025).

